WeightControl.com: What are the main findings?
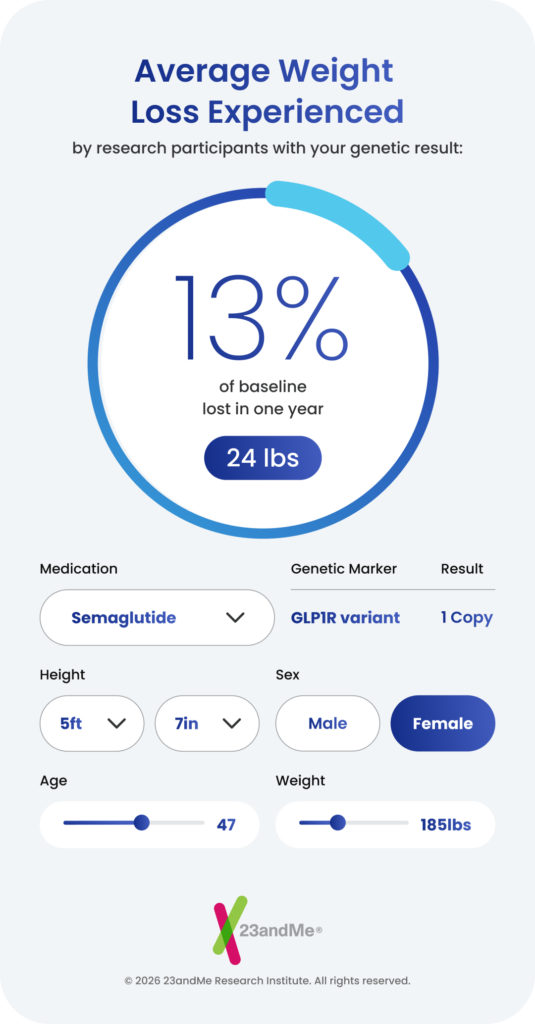
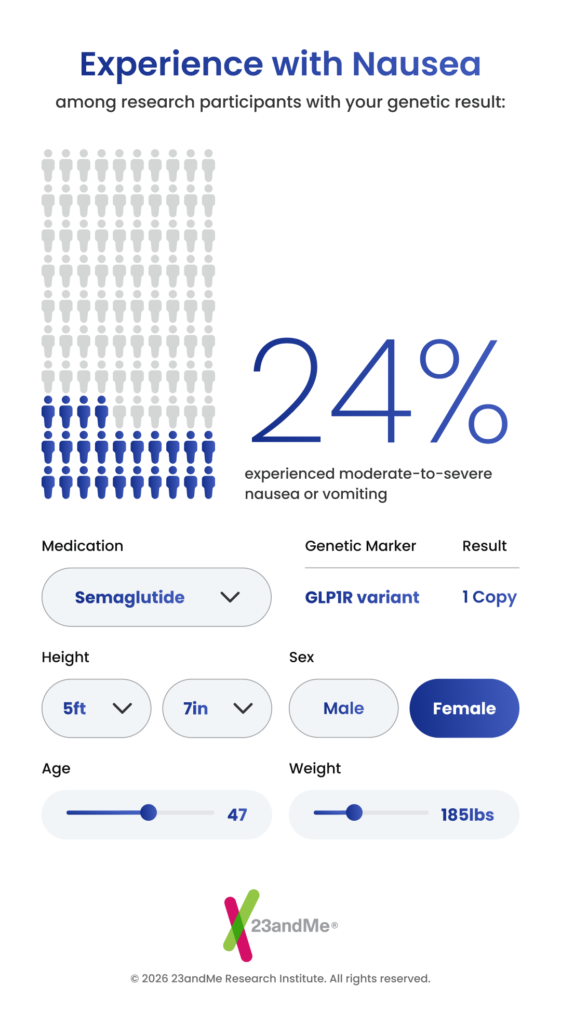
Response: We surveyed nearly 28,000 23andMe research participants who had used these medications and combined their treatment experiences with their genetic data. We found that a variant in the GLP-1 receptor gene – the protein these drugs directly target – is associated with greater weight loss. Each copy of this variant corresponds to roughly three quarters of a kilogram of additional weight loss. We also found that the same genetic region is linked to increased nausea and vomiting, suggesting that greater efficacy and gastrointestinal side effects may be two sides of the same coin. A second variant, in the GIP receptor gene, was associated with increased nausea and vomiting specifically in people taking tirzepatide (aka Mounjaro, Zepbound). Both findings point directly to the drug targets themselves, which gives us strong confidence they reflect real biology.

WeightControl.com: Is there a difference among the different meds?
Yes, and one of our most interesting findings highlights exactly that. The variant we identified in the GIP receptor gene is associated with increased nausea and vomiting only in people taking tirzepatide (aka Mounjaro, Zepbound), and not semaglutide (Ozempic, Wegovy). This makes perfect biological sense because tirzepatide targets both the GLP-1 and GIP receptors, while semaglutide only targets the GLP-1 receptor. We believe the GIP receptor normally helps buffer the nausea caused by GLP-1 activity, and that this variant reduces that protective effect. So for people carrying this variant, tirzepatide may come with a higher risk of gastrointestinal side effects than semaglutide, which is exactly the kind of information that could help a doctor choose between the two drugs for a particular patient.
WeightControl.com: What should readers take away from your report?
Response: The main takeaway is that your genetics can influence how well these medications work for you and what side effects you might experience. That’s not something most people think about when they start a GLP-1 medication, but our study shows it’s a real factor.
The broader message is that we’re moving toward a more personalized approach to obesity treatment. Rather than one-size-fits-all prescribing, we’re building the foundation for a future where a doctor can look at your genetic profile alongside your clinical history and make a more informed decision about which drug to prescribe, what dose to start with, and what to expect along the way. In fact, we’re already putting this into practice. Our findings are now part of a report available through 23andMe’s Total Health platform, a clinician-ordered service, so patients and their doctors can begin using this information today.
WeightControl.com: What recommendations do you have for future research as a result of this work?
Response: The variants we’ve identified are just the beginning. Larger and more diverse studies will almost certainly uncover additional genetic factors, and each one will improve our ability to predict treatment response.
There are also open questions regarding the influence of genetics on GLP-1 treatment. For example, it is known that when people stop taking these drugs, some people keep the weight off, whereas other revert to their pre-treatment weight. Is there a genetic component to that? We don’t yet know.
WeightControl.com: Is there anything else you would like to add? Any disclosures?
Response: I’d like to acknowledge the nearly 28,000 23andMe research participants who made this study possible by sharing their experiences with GLP-1 medications. Studies like this simply aren’t possible without people volunteering their data for research, and their contribution is advancing our understanding of these medications for everyone.
Interesting facts about 23andMe Research:
- 23andMe currently has more than 14 million genotyped members
- More than 80% genotyped members have consented to participate in research
- We are the world’s largest recontactable genetics research community, comprising more than 11 million consented and genotyped participants.
- 4B+ phenotypic data points collected to date
- While the size of the data set is impressive, it’s the recontactable nature that makes it exceptionally unique because researchers can gather data across a broader spectrum of behaviors and time frames than typically possible in traditional clinical settings.
- The average 23andMe research participant has informed more than 200 studies
- 23andMe published its first peer-reviewed paper in 2010.
- We now have 300+ peer-reviewed papers covering thousands of new genetic associations.
- Many of our studies were the largest of their kind, offering insights into conditions such as depression, Parkinson’s Disease, COVID-19, lung cancer, or rare diseases that are challenging to study, as well as breakthroughs in understanding human traits like height, genetic ancestry, and sexual orientation.
Citation:
Su, Q.J., Ashenhurst, J.R., Xu, W. et al. Genetic predictors of GLP1 receptor agonist weight loss and side effects. Nature (2026). https://doi.org/10.1038/s41586-026-10330-z
https://www.nature.com/articles/s41586-026-10330-z#citeas
The information on WeightControl.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.
Last Updated on May 7, 2026 by weightcontrol
